Comparison of Rapid Patient Cooling Methods
Robert B. Schock, Ph.D.
From the Department of Research and Development, The Sid Wolvek Research Center, Life Recovery Systems HD LLC, Kinnelon, NJ USA
As the field of therapeutic hypothermia is gaining interest, the variables which contribute to optimal results are becoming more apparent. One of these variables is the rapidity of cooling process. Newly published evidence supports the use of rapid cooling strategies . This report will review several such methods and discuss the advantages and disadvantages of each.
APPLICATION OF ICE TO THE SKIN
Direct cutaneous application of ice in crushed or cubed form, or incorporated into specially designed pads or blankets, is a seemingly straightforward means to achieve cooling induction. However, some have reported skin injuries resulting from the use of ice2, 3. Â A frostbite injury which resulted from the use of ice in the induction of therapeutic hypothermia in a patient is shown in Figure 1 below.
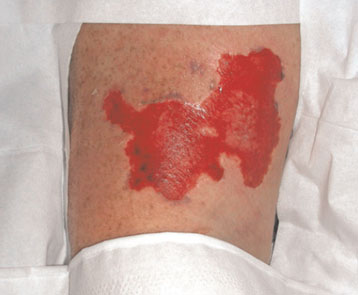
Figure 1: Frostbite injury resulting from the use of ice in the induction of therapeutic hypothermia2 (after de-roofing the blister).
Thus far only three clinical studies have been published in which patients were prospectively randomized to receive intravenous cold infusions in the field6, 8, 9.  None of these trials have proven a long-term benefit to this approach in comparison with in-hospital cooling alone. In fact, the largest of these trials, which enrolled 234 patients, observed a trend for worsened outcomes with patients cooled by paramedics using iced intravenous infusion, in comparison with the use of in-hospital cooling alone6 (see Figure 3 below):
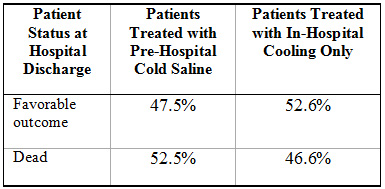
Figure 3: Trend for unfavorable outcomes with use of pre-hospital cold intravenous infusion, observed in 234-patient randomized study (Bernard SA et al, Circulation 2010)6
The poor outcomes in the randomized studies of cold intravenous infusion may be due to one or more of the following factors:
- The additional 2L of intravenous fluid may add undesirably to cardiac pre-load, which might not be tolerated by some patients and may add to damage of the heart10.
- Cold infusion typically just cools patients to a level between 34°C and 35°C6. This puts them in the zone of maximal thermoregulatory response11. Shivering and non-shivering thermogenesis can add tremendous stresses to the patient. Shivering typically continues until patients are below 33.5°C11.
- Patients cooled with cold infusion and left lingering in the 34°C to 35°C range tend to rewarm themselves6, 7. This requires that they expend precious energy, adding to overall physiological stresses. If they are subjected to a second cooling induction to get below a target of 34°C, they must again pass through the zone of maximal thermoregulatory response – a further source of stress.
Perhaps because of the above negative consequences of cold intravenous infusions, a number of studies in which this cooling method has been used have failed to demonstrate benefits of earlier cooling6, 9, 12, 13. Conversely, the studies most strongly supporting the use of earlier induction of therapeutic hypothermia have avoided the use of cold infusions1, 14, 15.
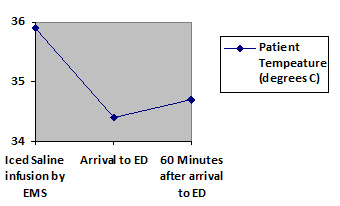
The Association of Chartered Physiotherapists in Sports Medicine published guidelines on the application of ice for soft tissue injuries4 which provide the following recommendations:(1) The most effective duration of application is 20–30 minutes, with a maximum safe period of 30 minutes.(2) A damp towel should be placed between the cooling agent and the skin.(3) Care should be taken with the application of ice (or cooling agent) on areas with little subcutaneous fat or muscle, and in the region of superficial nerves, with a maximum cooling period of 10 minutes.Infusion of Large Volume Ice-Cold Crystalloid FluidThis is a relatively simple method that requires little expense, and it can be started in the pre-hospital setting5. Usually only 1.5°C of patient temperature reduction can be provided with this method, and patients have a tendency to rewarm themselves soon after the infusion is stopped6, 7 (see Figure 2)
LIFE RECOVERY SYSTEMS THERMOSUIT®
This cooling method utilizes the principal of ice water immersion of the patient in a low-volume disposable enclosure16. This enables patients to be safely and rapidly cooled to 33°C with a typical treatment period of just 30 minutes. This minimizes the time that the patient remains in the stressful zone of maximal thermoregulatory response. As the patient is quickly and reliably cooled below the 33.5°C shivering threshold, he can then be removed from the device and remain cold for hours without further cooling, (see Figure 4):
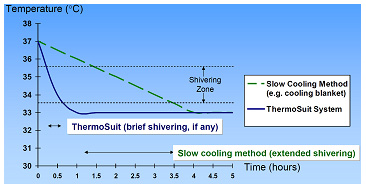
Figure 4: Temperature vs. Time for patient cooled by ThermoSuit System, compared to slower cooling method. The rapid cooling of the ThermoSuit System minimizes the duration of patient exposure to stressful thermoregulatory responses.
The ThermoSuit approach has never been associated with any reports of skin injury. This is a result of the product design:
- The water used is above the freezing point, preventing the development of frostbite.
- The ThermoSuit does not have any adhesive surfaces to injure the skin.
- A soft, low-shear batting layer protects the skin from abrasions and pressure sores.
The ThermoSuit System has been demonstrated to provide safe and rapid cooling in independent studies of therapeutic hypothermia16, 17, 18. It does not carry the risk of skin injury associated with ice pack cooling, nor the excess ventricular pre-load associated with high volume intravenous infusions. Its rapid cooling rate minimizes the time during which the patient is subjected to the stresses of thermoregulatory responses to cooling. Because of its excellent record of safety and cooling performance, it is expected that this approach will be increasingly adopted as the method of choice for noninvasive induction of therapeutic hypothermia.
References
[1] Sendelbach S, Hearst MO, Johnson PJ, Unger BT, Mooney MR, “Effects of Variation in Temperature Management on Cerebral Performance Category Scores in Patients Who Received Therapeutic Hypothermia Post Cardiac Arrestâ€. Resuscitation 83 (2012), 829-834.
[2] Graham CA, Stevenson J, “Frozen chips: an unusual cause of severe frostbite injuryâ€, Br J Sports Med; 34:382-384.
[3] Lohan P, Hart A, “Therapeutic hypothermia and frostbite injury: a preventable sourceâ€, Scottish Medical Journal 2011; 56: 1-2.
[4] Kerr KM, Daley L, Booth L, et al. Guidelines for the management of soft tissue (musculoskeletal) injury with Protection, Rest, Ice, Compression and Elevation (PRICE) during the first 72 hours. London: Chartered Society of Physiotherapy, 1999.
[5] Arulkumaran N et al, “Use of ice-cold crystalloid for inducing mild therapeutic hypothermia following out-of-hospital cardiac arrestâ€, Resuscitation 83(2012) 151-158.
[6] Bernard S, Smith K, Cameron P, et al. Induction of therapeutic hypothermia by paramedics after resuscitation from out-of-hospital ventricular fibrillation cardiac arrest. A randomized controlled trial. Circulation. 2010; 122:737-742.
[7] Kliegel A et al, “Intravenous Administration of Cold Fluids, Sedation, Analgesia and Muscle Relaxation if Sufficient for Induction but Not for Maintenance of Therapeutic Hypothermia after Cardiac Arrestâ€, ERC 2006 Proceedings, Resuscitation April 2006, p. 58.
[8] Kim F et al, “Pilot Randomized Clinical Trial of Prehospital Induction of Mild Hypothermia in Out of Hospital Cardiac Arrest Patients with a Rapid Infusion of 4° Normal Salineâ€, Circulation 2007; 115:4064-70.
[9] Kamarainen A et al, “Prehospital therapeutic hypothermia for comatose survivors of cardiac arrest: a randomized controlled trialâ€, Acta Anaesthesiol Scand 2009; 53: 900-907.
[10] Yannopoulos D et al, “Intra-Cardiopulmonary Resuscitation Hypothermia With and Without Volume Loading in and Ischemic Model of Cardiac Arrestâ€, Circulation 2009, 120: 1426-1435.
[11] Polderman K and Herold I, “Therapeutic hypothermia and controlled normothermia In the intensive care unit: Practical considerations, side effects, and cooling methodsâ€, Crit Care Med 2009; 1101-1120.
[12] Nielsen N et al, “Outcome, timing, and adverse events in therapeutic hypothermia after out-of-hospital cardiac arrestâ€, Acta Anesthesiol Scand 2009; 53: 926-934.
[13] Alongi S et al, “Early-versus late initiation of therapeutic hypothermia after cardiac arrest: Preliminary observations from the experience of 17 Italian intensive care unitsâ€, Resuscitation 83 (2012) 823-828.
[14]Wolff B, Machill K, Schumacher D, Schulzki I, Werner D, “Early achievement of mild therapeutic hypothermia and the neurologic outcome after cardiac arrestâ€, International Journal of Cardiology 2009.
[15] Castrén M, Nordberg P, Svensson L, et al. Intra-arrest transnasal evaporative cooling. A randomized, prehospital, multicenter study (PRINCE: Pre-ROSC IntraNasal Cooling Effectiveness). Circulation. 2010; 122:729-736.
[16] Howes D, Ohley W, Dorian P, Klock JC, Freedman R, Schock R, Krizanac D, Holzer M, “Rapid Induction of Therapeutic Hypothermia using Convective-Immersion Surface Cooling: Safety, Efficacy, and Outcomesâ€, Resuscitation 2010 Vol. 81, no .4, 388-392.
[17] McMullan P, Howes D, Ohley W, Dorian R, Freedman R, Schock RB, Holzer M, “41 – Liquid Convection Surface Cooling Leads to High Rate of Recovery of Post-VT/VF Patientsâ€, Circulation 2011 (Abstract);124:A41.
[18] Kudagi VS, McMullan PW, Jr., White CJ, “Rapid induction of therapeutic hypothermia in comatose survivors of cardiac arrest using liquid-convection immersion cooling”, Society for Cardiovascular Angiography and Interventions 2012 (Abstract).