
 |
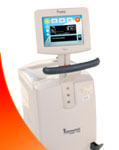
FOR IMMEDIATE RELEASE Monday, June 12, 2023 LIFE RECOVERY SYSTEMS AND FDA AGREE TO CONCLUDE PILOT STROKE COOLING STUDY AND MOVE FORWARD TO RANDOMIZED TRIAL Life Recovery Systems (LRS) has received concurrence from the US Food and Drug Administration (FDA) to successfully end its pilot study (SISCO) with the ThermoSuit® System in the treatment of ischemic stroke. The FDA met with LRS on June 1, 2023, to review the SISCO study results, concluding that that the TSS had achieved its objectives of feasibility, safety, and rapid cooling, while showing a very strong trend for brain protection and better recovery. FDA recommended that LRS end the SISCO study and move forward with a prospective randomized trial of rapid cooling in acute ischemic strokes. According to Dr. Robert Schock, VP of R&D at LRS, “We’re most pleased with the excellent results in the SISCO Study. Extensive data from laboratory studies have long predicted that rapid cooling methods would be highly effective in reducing brain injuries in acute ischemic strokes, while the use of slower, weaker cooling methods have had disappointing results. In contrast the TSS showed that rapid non-invasive cooling was safe and effective. The support of the FDA allowing us to proceed with the definitive prospective randomized study reflects the clinical potential of the ThermoSuit system. With 800,000 strokes/year in the US, the TSS has the potential to improve clinical outcomes while significantly decreasing the cost of treatment.” The ThermoSuit System uniquely uses direct liquid contact and convective heat transfer to rapidly reduce core body temperature. Core temperature is monitored throughout cooling, and approaching target temperature the iced water is removed from the suit. The TSS is then removed from the patient, who will stay stable in the target range for hours, allowing transfer for diagnostic and therapeutic procedures unencumbered. The TSS is easily deployed, usually cooling to target in 40 minutes or less. Matt Center, LRS CEO, notes, “This recognition by the FDA is a testament to the unmatched cooling power of the ThermoSuit system. This greatly enhances the investment opportunity in LRS and we are looking forward to continuing to aggressively pursue the FDA clearance for the stroke market.” About Life Recovery Systems Founded in 2003, Life Recovery Systems focuses on ground-breaking, innovative medical devices for the emergency medical care market. Its mission is to save and preserve the quality of patients’ lives. LRS’ first product, the TSS, was developed with funding assistance of NIH NHLBI to provide medical personnel with a safe, rapid method for cooling patients. LRS has offices in Alexandria, LA and Kinnelon, NJ, with the Sid Wolvek Research Center also based in NJ. Contact: Robert Schock For investment inquiries please contact Matt Center |
|
| Life Recovery Systems Video | ||
