Scientific Presentation
Presented Feb. 7-9, 2024
Therapeutic Hypothermia With Thin Liquid Convection is Safe and Feasible in Acute Stroke Patients: The Helping Stroke Patients With ThermoSuit Cooling (SISCO) Pilot Study, accepted as Abstract #3695 at the 2024 International Stroke Conference, Phoenix, AZ.
(Note: this presentation will discuss clinical testing conducted under an Investigational Device Exemption in ischemic stroke patients. This indication is not yet approved by FDA for the ThermoSuit.)
About Life Recovery Systems
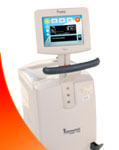
Founded in 2003, Life Recovery Systems focuses on ground breaking, innovative medical devices for the emergency medical care market. Its mission is to save and preserve the quality of lives. Its first product, the ThermoSuit System, was developed with the assistance of NIH NHLBI funding to provide physicians and nurses with a safe, convenient, and extremely rapid method for cooling patients. It has received FDA 510(k) clearance for temperature reduction in patients where clinically indicated, e.g. in hyperthermic patients. It won a Medical Design Excellence Award in 2009, and multicenter clinical results were published in the Journal Resuscitation in 2010. Headquartered in Kinnelon, NJ, Life Recovery Systems also has offices in Alexandria, LA. https://www.life-recovery.com.
Contact: Robert Schock
info@life-recovery.com
Phone: 973-283-2800